In the present thesis emphasis has been given to study
free radical reactions in different environment with a variety of
bio-molecules to address the aforesaid queries. After a brief introduction
and experimental description, the research results start with the
redox reaction of bilirubin (BR), a good heme model, in micelles.
The hydrated electrons have been seen to react with BR four times
faster as compared to that in the pure homogeneous aqueous solution.
It has also been observed that reduction reaction of BR by CO2.-
radical that could not be observed in the aqueous solution proceeded
with almost diffusion-controlled rate in a cationic micelle. The reaction
has been explained on the basis of the effect of charged surface on
the micelle, which provides the catalytic environment for electron
transfer to occur which otherwise was not possible. The reaction of
BR has been extended in bovine serum albumin (BSA) solution (the protein
is used as the biological microheterogeneous medium). It has been
observed that BSA protects the bound BR molecule from oxidative attack
by free radicals like .OH, CCl3OO.,
N3. etc. It has been concluded that biological
microheterogeneous environment can induce even negative catalytic
effect for the destruction of bio-molecules by different free radicals,
which can otherwise complicate the situation.
In addition to these examples it can be mentioned here
in short that while studying the eaq- reaction
with different metal ions in water-in-oil microemulsion it has been
observed that microemulsion can provide novel catalytic media for
synthesizing monodispersed metal nano particles.
To address the second point, a water-in-oil microemulsion
has been used. The microemulsion is composed of NaLS/water/cyclohexane/1-pentanol.
In a microemulsion, there are two sources of hydrated electrons; one
is the scavenging of excess electrons produced in the hydrocarbon
phase by the water pools and the other is the direct radiolysis of
water. Remarkably high lifetime (20 ms)
for hydrated electrons has been obtained. In general, these are two
orders of magnitude higher than those reported earlier in reverse
micelles. The decay kinetics of hydrated electrons has been employed
to determine the water pool size and location of the probes. The variation
of hydrated electron concentration with time is given by:
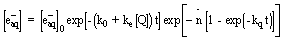 |
(1) |
Where [eaq-] and [eaq-]0
are the hydrated electron concentrations at time t and zero, respectively.
k0 is the first order decay rate constant of the hydrated
electron in the absence of solutes, ke is the bimolecular
rate constant for an exchange process that involves water pool collisions,
kq is the hydrated electron decay constant in the presence
of solutes and
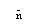 |
is the average number of solutes per micelle. |
At any given time t, P0, the probability
of finding zero solute per micelle is given by
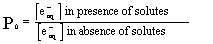 |
(2) |
Then following the Poisson distribution,
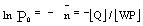 |
(3) |
Where [Q] and [WP] are the concentration of the solute
and water pool, respectively. Hence a plot of ln P0 versus
[Q] gives a measure of the water pool concentration, which in turn
gives the radius of the water pools, assuming these are of spherical
shape. In our experiment we have measured the water core radii from
the electron decay in presence and absence of solute at a particular
time window. Different solutes used for this purpose are CuSO4,
N,N-dimethylformamide, CCl4 and BSA. Following the procedure
described, water core radii have been determined for almost all w0
(w0 = [water] / [surfactant]) values using different solutes
as mentioned. Earlier an empirical relation between w0
and r as r = 1.5 w0 had been proposed. In our all measurements
the radii of the water pool are close to 1.5w0.
Further, as the absorbance of the hydrated electrons
is directly proportional to the concentration of eaq-,
equation [1] reduces to
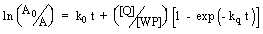 |
(4) |
where A0 and A are the absorbances of hydrated
electrons at time 0 and time t, respectively. kq has been
evaluated after modifying equation 4 and by plotting ln (A'0/A')
versus [Q], Where A'0 and A' are the absorbances of hydrated
electrons at time t in absence and presence of quenchers, respectively.
As kq varies as 1/wo3 when solute
is located in water pool and varies as 1/wo2
when solute is located at the interface we have determined the location
of different solute in the microemulsion by following the decay kinetics
of the hydrated electrons. Hence it has been concluded that radiation
chemistry may be employed to determine the physical parameters of
a microemulsion.
Third point is to evaluate whether the microheterogeneous
media can really provide a bio-mimicking environment. For this purpose
the aforesaid microemulsion has been chosen as the model. The exact
role of b-carotene in cancer prevention
is still speculative although it is known as an ubiquitous free radical
quencher whereas retinol has drawn extensive attention for its cancer
prevention activity. b-carotene is known
as provitamin A due to its conversion into retinal by the enzyme
dioxygenase via its central or excentric cleavage. This has been demonstrated
earlier by in vivo tests with vitamin A deficient animals. Although
the enzyme is present chiefly in the intestine and possibly in the
liver, accumulation of vitamin A has been surprisingly noticed in
many tissues in mice. This leads to the speculation as to whether
it is possible that vitamin A also be derived from b-carotene
by some purely chemical protocol. This question has been addressed
by focusing investigation on the interaction of b-carotene
with reactive oxygen species in the microemulsion. Other than the
time-resolved study, steady-state analysis of the reaction products
showed the presence of retinol. UV-Vis absorption and fluorescence
study on the product gave the information that retinol had been formed
along with other products in the reaction of CCl3OO.
radical with b-carotene in microemulsion.
HPLC analysis of the products has confirmed that around 18% retinol
was present. This might also explain the presence of retinol in organs
other than liver as found in mice earlier. It should be mentioned
that in earlier radiation chemical studies on b-carotene
in homogeneous solution and in micelles, formation of vitamin A had
not been observed. Hence, it can be concluded that in some cases the
microemulsion can really mimic the physiological situation. In recent
times, microemulsions are being widely used for biochemical studies.
Still there is ample scope for future researchers to study oxidation
reactions in microemulsions, though several studies are in progress
in other model systems such as liposome, microsome, etc.

