 |
 |
 |
 |
|
Chemical Education International, Vol. 3, No. 1, AN-8, Received November 1, 2002
|
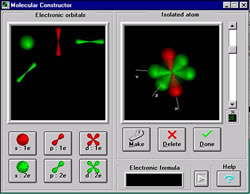 |
| Figure 1. In this design studio students select orbitals from a kit and use them to design and construct atoms and molecules. The computer provides appropriate feedback to students on their designs. (Larger View) |
The design process requires students to understand the content deeply, to formulate hypotheses, and to use chemical laws, theories, and rules. Instead of consulting a textbook, students find the information they need through links in the web pages. The course is matched to the provisions and requirements of the National Science Education Standards for teaching, content, and assessment.
The Quests can be completed in almost any order and some teachers have used them as supplements to a more traditional, textbook-based course. However, teachers who completed them in the order given in Table 1 felt that by working from the smallest to larger particles students learn how the properties of a bulk sample of matter arise from the properties of the atoms and molecules that make up the sample.
Table 1: The Eight Quests of ChemDiscovery
|
Quest |
Content |
Motivation |
|
1 |
To design atomic nuclei and atoms To design a set of atomic nuclei To design electronic structures |
Origin of the universe The Sun Radioactive isotopes in everyday life |
|
2 |
To design electronic structures of atoms |
Starlight History of science |
|
3 |
To design models of monatomic ions |
Ions in the plasma in outer space Ions in food, glass, seawater |
|
4 |
To design elemental substances Gaseous elements Liquid substances Solid substances: Elemental crystals |
Stars and planets (core, crust, and atmosphere) Air pollution, solar cells, automobiles |
|
5 |
To
design chemical reactions between |
Atmosphere, hydrosphere, and geosphere of Earth Corrosion |
|
6 |
To design models of binary compounds Formulas and bonding Basic molecular shapes Basic types of crystal packings |
The water, carbon, and nitrogen cycles |
|
7 |
To design and explore systems: Solutions |
The water cycle and water pollution Water treatment |
|
8 |
To investigate and design a system: Chemical reactions Stoichiometry Kinetics Equilibrium Thermochemistry |
Ecology Metabolic cycles |
ChemDiscovery is delivered on a CD-ROM with a teacher guide and two accompanying books: a student guide, and a hands-on laboratory manual. In addition, the teacher CD-ROM contains detailed information on the Quests, solutions to assignments, and photocopy masters for worksheets and assessments.
3. How ChemDiscovery Works
Each Quest formulates learning goals that students can meet directly through the activities or by first exploring one of two contextual motivation tools: Design of the Universe and/or Living in the Universe. These tools allow students to enter the world of chemistry from environmental, scientific, and social perspectives. Because students choose their starting point and design unique pathways through the learning environment, a teacher can accommodate different learning styles and different levels of difficulty in the same classroom. A field work guide shows teachers how to introduce field experiences that relate the principles they are learning to their lives.
Design activities are not ordinarily encountered by secondary school students (6). Yet design processes play important roles in our daily lives as well as in science and engineering (7,8). The ChemDiscovery design activities involve students in understanding needs and responsibilities, selecting raw materials, checking databases, predicting the properties of objects (nuclei, atoms, ions, molecules, crystals, and larger systems), designing them, and evaluating their predictions by performing hands-on laboratory experiments. As they carry out their design projects, students become engaged in authentic scientific practices. That is, students using ChemDiscovery work like scientists with the aid of course learning tools (Table 2).
Table 2: Learning Tools Provided by the ChemDiscovery Learning Environment
| These Inquiry Steps | Use These Supported Learning Tools |
| Observe phenomena and formulate problems |
The environment Hands-on labs Video labs |
| Search for information and choose materials | Resources and databases |
| Design, model, and construct chemical objects or phenomena | Design studios |
| Analyze structures and predict properties |
Interactive computer activities Worksheets |
| Check predictions and make discoveries in the laboratory |
Computer feedback Hands-on labs Video labs |
| Assume responsibility for the environment |
Living in the Universe Field work |
ChemDiscovery assessments include both traditional tests and evaluations of inquiry skills. For example, the assessments require students to design, model, and construct chemical objects and phenomena. They also evaluate student ability to use professional scientific databases, the same type used by scientists.
4. A Typical Class
A day in a typical ChemDiscovery classroom begins when the teacher introduces the learning objectives for a topic and relates them to previous work. She then gives each student a navigational checklist to fill in while completing the activities. Each group of students maps out a learning pathway and begins to work the hands-on lab or to solve the problems presented by the computer. Some of the activities are completed on the computer and the results are printed for the teacher to review. Others are completed on worksheets using the resources and databases on the computer.
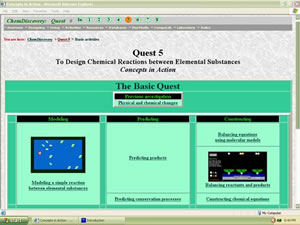 |
| Figure 2. This menu introduces students to a series of design and prediction activities. (Larger View) |
For example, in Quest 5 (Fig. 2), students use periodic table databases to predict the formulas of the products formed when two elements react. They enter formulas on a computer web page and receive feedback on their predictions from the computer. In the design studio students model chemical equations for reactions between the elements (Fig. 3). They then use model kits along with a worksheet to complete molecular-level drawings of chemical reactions. After students have worked on their assignments for a while the teacher reviews the main points of the lesson. A final Quest 5 assignment may involve a fieldwork activity in which students find examples of chemical reactions such as corrosion in their neighborhoods.
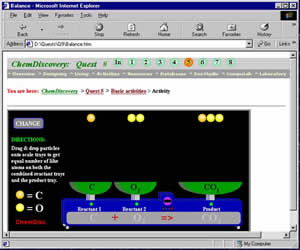 |
| Figure 3. Students use this design studio to model chemical reactions between the elements. (Larger View) |
5. The Classroom Impact
During the field testing of the ChemDiscovery curriculum three aspects of learning chemistry were investigated: the level to which teachers adapted to the new approach, the barriers to implementation, and changes in classroom climate and interactions. Teachers in several of the classrooms were observed using ChemDiscovery and students and teachers in all classrooms kept daily journals in which they reported their experiences and concerns.
Traditional as well as technical barriers are commonly encountered when teachers introduce computer-based chemistry experiences. Therefore, it was not surprising that most of the field-test teachers needed help in locating enough computers to run the curriculum. The State of Colorado was approached to inquire whether the state would be willing to provide computers for ten classrooms. The classrooms would then serve as model science technology classrooms and the teachers would serve as technology experts in their districts. The state agreed and once the computers were in place, the hardware problems encountered implementing the curriculum were mainly associated with the local area network configurations in the schools. In general, teachers found that setting up their computer classrooms took longer than they had anticipated and in some cases, the computers were not even delivered by the beginning of classes. However, eventually all the teachers were running the software. As with any new approach, teachers found they needed to allow extra time for preparation and for dealing with unexpected problems.
Observers noted that the integral use of technology in the classroom changed the focus of student-teacher interactions from a teacher-led lecture format to one in which students spent more time working cooperatively in groups and teachers spent more time acting as facilitators (9). During the field test of the curriculum two of the teachers were observed on a regular basis and the time spent by teachers and students on different classroom activities was noted (10). Each teacher taught one class in a traditional manner, without the use of computers, and one in which the class used ChemDiscovery as the primary curriculum. In the ChemDiscovery classrooms the teachers spent about the same amount of time facilitating independent student work (38%) as they did lecturing (40%). However, in classrooms where they used traditional teaching methods the teachers spent much more time lecturing (58%) than they did facilitating (13%). All the teachers alternated the facilitator role with more traditional roles, depending on the classroom activity. The degree to which a teacher played a facilitator role depended on the degree to which the teacher was committed to student-centered learning. The extent to which lectures and textbooks were used was also related to the degree to which the teacher was committed to independent learning. Teachers who were fully committed to independent learning were more likely to use the curriculum as intended. Those more comfortable with traditional, lecture-based methods of teaching were more likely to add lectures or textbooks.
Learning to teach in accord with the science teaching standards, which were new at the time, especially learning to teach via inquiry, required additional training and ongoing support for the teachers. However, all the teachers felt that they had learned new skills. One teacher reported (11):
The presence of computers in my classroom has dramatically altered my approach to instruction, enhanced my understanding of my instructional areas, re-energized my instruction, and changed my professional goals. I was a classroom teacher with 21 years of successful experience but I was preparing to become an administrator, in part because the classroom had lost its challenge. The addition of computers to my classroom caused me to wonder exactly how this new technology would mesh with what I knew so well. What has evolved is a dynamic learning environment in which both instructor and student continuously refine their knowledge base through web access. The availability of world-wide access to informational resources has caused me to restructure my course work so that informational support of content is not only text based, but also web based.
One of the biggest differences between a ChemDiscovery classroom and a traditional classroom is the extent to which students learn independently. Students who were used to more traditional learning environments, with textbooks and lectures, found independent learning a challenge and needed more guidance to begin. However, despite the initial resistance from some students, teachers found advantages to the active learning approach. For example, one teacher claimed that her students had learned the material to such a depth that they no longer needed to cram for exams (9):
It became their information instead of information that they were cramming, you know, from a book. It was inside of them. They had the pieces. And they had the understanding without trying to, they have the puzzle put together. There was much more coherent, cohesive understanding of how it all goes together.
Another teacher reported (11),
The students have had to learn how to learn in a whole new way. They are no longer given the knowledge that I think is important but have to search and research for themselves. Making critical decisions of this kind has certainly stretched and frustrated them. I think that they have become better learners in the long run.
6. Conclusions
Previous studies have shown that students are willing to learn with new technologies and find unique advantages to learning with molecular modeling tools and simulations when they have control over the modeling and simulation process (12,13,14,15). The findings of the field test of this new approach to teaching chemistry are consistent with those studies. A primary goal of the field test evaluation was to discover whether using a computer-centered inquiry curriculum fosters independent student activities. Both observer reports and teacher journals imply that the students were not only working independently, but that they may have been becoming more successful learners. Students successful at learning use more active learning strategies than those used by immature learners (16). For example, they connect new knowledge to what they already know, they organize and review their knowledge and monitor their understanding, while immature learners use more passive learning strategies. This study suggests that interactive multimedia courseware may be able to help teachers to provide a learning environment that encourages the development of active learning strategies by requiring students to learn independently.
7. Acknowledgements
The authors would like to thank Ron Anderson, University of Colorado at Boulder, and his former students Cory Buxton and Megan Mistler-Jackson, for their insightful evaluation of ChemDiscovery classrooms, and Richard Mayer, University of California at Santa Barbara, for helpful comments on this article. The authors would also like to acknowledge the support of the National Science Foundation Division of Elementary, Secondary, and Informal Science Education, for their funding of the development and evaluation of ChemDiscovery through Project #ESI-9550545. Additional funding was provided by the Soros Foundation and by the Technology Learning Grant and Revolving Loan Program of the State of Colorado Department of Higher Education.
References
(1) Mayer, E.R., The Promise of Educational Psychology, V. 2; Prentice-Hall: Upper Saddle River, NJ, 2002.
(2) Agapova, O., Jones, L, & Ushakov, A., ChemDiscovery, Kendall-Hunt: Dubuque, IA, 2002: http://www.chemdiscovery.com
(3) Agapova, O., Jones, L., and Ushakov, A., Informatika I Obrazovanie, 1, 105-109, 1996.
(4) National Research Council, National Science Education Standards. National Academy Press. Washington D.C., 1996. Available at: http://www.nap.edu/readingroom/books/nses/html/
(5) Jones, L, Jordan, K., and Stillings, N., Molecular Visualization in Science Education: Report from the Molecular Visualization in Science Education Workshop (2001): http://pro3.chem.pitt.edu/workshop/workshop_report_180701.pdf
(6) Jones, L. L., Uniserve News, 14, November, 1999: http://science.uniserve.edu.au/newsletter/vol14/jones.html.
(7) Agapova, O.I., Ushakov, A.S., Think, 12, 7-9, 1998.
(8) Agapova, O.I., Ushakov, A.S., Technos, 8, (1), 27-31, 1999.
(9) Anderson, R., Buxton, C. and Mistler-Jackson, M., Evaluation of the ChemQuest program in the context of the third year field test. National Science Foundation (1999)
(10) Schoenfeld-Tacher, R., Madden, S., Pentecost, T., Mecklin, C. and Jones, L., A Systematic Comparison of Technology-Based and Traditional High School Chemistry Classrooms, National Meeting of the National Association for Research in Science Teaching, Boston, MA, March 30, 1999.
(11) Jones, L. L., Technology Excellence in Learning Award Final Report: Technology-based Model Science Classrooms. Colorado Commission on Higher Education (1999).
(12)
Jones, L. L., New Initiatives in Chemical Education (Summer,
1996): http://www.inform.umd.edu:8080/EdRes/Topic/Chemistry/
ChemConference/ChemConf96/Jones/Paper3.html
(14) Dori, Y. J., and Barak, M. Educational Technology and Society, 4, (1), 61-73, 2001.
(15) Jones, L. L. and Smith, S. G., Pure and Applied Chemistry, 65, 245-249, 1993.
(16) Friedler, Y., Nachmias, R., and Songer, N. B., School Science and Mathematics, 89, 58-67, 1989.
Posted February 25, 2003.
Last modified 20.07.03
| CEI is a newsletter of the Committee on Chemistry Education (CCE) of IUPAC |
 |
 |
 |
 |
