Ph.D. Thesis
Title On-line Concentration of Analytes in Electrokinetic
Chromatography
Advisers Prof. Shigeru Terabe
Thesis Committee Prof. Shinichi Nakatsuji, Department of Material
Science, Faculty of Science; Prof. Tadashi Okuyama, Department of
Material Science, Faculty of Science; Prof. Takashi Iyanagi, Department
of Life Science, Faculty of Science; Assoc. Prof. Koji Otsuka, Department
of Material Science, Faculty of Science; Prof. Takeshi Hirokawa, Department
of Applied Physics and Chemistry, Faculty of Engineering, Hiroshima
University.
Essay
Scientists in various fields have been delighted at
the ability of electrokinetic chromatography (EKC) - a mode of capillary
electrophoresis (CE) - to solve many chemical analysis problems and
to complement other separation methods like gas chromatography and
high performance liquid chromatography. Applications are in the areas
of toxicology, environmental pollution, pharmacokinetics, clinical
diagnosis, cell composition, to name a few. Notable aspects of EKC
include high efficiencies, technical simplicity, applicability to
most analytes, requires small amounts of samples and reagents, and
miniaturization (EKC on chips). The basic experimental set-up consists
of a narrow bore capillary (e.g., 50 mm i.d.), a voltage delivery
device (up to 30 kV), a detector equipped with data acquisition and
analysis tools, and sample and separation solution reservoirs. In
addition to the aqueous buffer used in free solution CE or capillary
zone electrophoresis (CZE), a major component called pseudostationary
phase (PS) is added. Separation principle in EKC is similar to that
of classical chromatography, which is analyte partitioning between
the PS (e.g., micelle) and the aqueous phase. The electrokinetic phenomenon,
including electrophoresis and electroosmosis, is the means of transporting
the PS and analytes inside the capillary. One great concern, however,
is the low concentration sensitivity in the ppm levels as a consequence
of the short optical pathlength for on-capillary photometric detection
and the small volume of sample that can be injected. This hinders
application to trace analysis without several time consuming off-line
pre-concentration steps.
Powerful laser induced fluorescence and electrochemical
detectors can improve detection sensitivity several orders but are
not widely applicable and very expensive for an ordinary laboratory.
A more practical and easy way is the 'chemical approach' or to concentrate
the analytes on-capillary or on-line. This is done by manipulating
the composition of the sample and background solutions together with
simple injection procedures without alteration of present commercial
instrumentation. For example, transient isotachophoresis and sample
stacking have provided at most hundred-fold increases in detector
response. Here, new and general approaches to concentrate analytes
in EKC inside the capillary are then presented. Detection sensitivity
with photometric detection was improved from ten to almost a million-fold.
The proposed techniques will eventually widen the applicability of
EKC in trace analysis. Likewise, sound understanding of the narrowing
mechanisms involved opened possibilities for elucidating the behavior
of analyte zones in CE.
A novel phenomenon termed as 'sweeping' was introduced
[J.P. Quirino, S. Terabe, Science 282, 465 (1998)] which, in theory,
predicts an almost unlimited improvement in detection sensitivity
for analytes having high affinities toward the PS: 5000-fold improvements
have been demonstrated experimentally (equation 1).
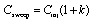 |
(1) |
Where Csweep and Cinj are the concentrations of the
analyte zones after sweeping and during sample introduction, respectively.
The retention factor, k, is directly related to the affinity of the
analyte toward the PS. The above equation is useful for both neutral
and charged analytes, making the sweeping technique a universal approach
to on-line concentration as long as the k is reasonably high. Sweeping
results when the PS, which penetrates the sample zone entraps and
accumulates the analytes. It is analogous to using a broom (PS) to
carefully carry along grains of rice (analyte) scattered on the floor.
The sample is prepared in a matrix that is free of the PS and has
a conductance similar to or higher than that of the background solution.
Environmentally and biologically important compounds such as phenoxyacid
herbicides, polycyclic aromatic hydrocarbons, monocyclic aromatic
amines, alkyl phenyl ketones, dialkyl phthalates, alkaloids, and steroids
have been successfully concentrated using micelles, microemulsions,
and a charged cyclodextrin as PS.
Sample stacking techniques for neutral analytes have
also been developed. Sample stacking, which was originally developed
for the on-line concentration of charged analytes in capillary zone
electrophoresis, is defined as the movement of ions across a concentration
boundary that separates regions of high and low field strengths. Since
the electrophoretic velocity in the high field strength zone (lower
conductivity sample zone) is higher compared to the low field zone
(higher conductivity separation zone), ions will slow down once it
reaches the boundary causing the narrowing of zones. Since neutral
analytes are unaffected by an enhanced field, sample stacking was
thought to be not amenable for neutral analytes in EKC. However, by
careful manipulation of sample matrix and some simple tricks, sample
stacking was applied to neutral analytes as well. In EKC, the PS will
provide the neutral analytes effective electrophoretic velocities
necessary for the approach. Many environmentally relevant phenols,
dioxins, alkylphenyl ketones, phenylurea herbicides, and steroids
have been successfully concentrated using monomeric and monomolecular
micelles. On top of this, sweeping and the different sample stacking
techniques developed were found to be orthogonal methods, the sample
stacking techniques are more useful for low to moderately high k
analytes while sweeping is for high to very high k analytes.
Finally, a novel method that combines two on-line concentration
techniques, namely, sample stacking with electrokinetic injection
in the CZE mode and sweeping, afforded the detection of positively
chargeable analytes in the parts per trillion levels with very high
average plate numbers (e.g., 4.4 x 105).
This is lowest concentration level reported by direct photometric
detection in CE, which translates to detection improvements approaching
a million-fold. The main idea is to selectively inject by electrokinetic
injection as many molecules as possible from a very dilute sample
solution. This creates long concentrated zones of cationic species,
which is then focused further by sweeping. Several drugs (e.g., b-adrenergic
blockers, tetracycline, and laudanosine), seven environmentally important
monocyclic aromatic amines, and two benzidines have been successfully
analyzed. At the end, simple, faster, sensitive, reproducible, and
cheap real-world trace analysis can be achieved by EKC with the help
of the above on-line concentration techniques.

