|
 |
 |
 |
|
Chemical Education International, Vol. 2, Issue 1, 6-13, Published in August 3, 2001
Zafra M. Lerman
English
dictionaries define visualizing as "forming a mental image
or vision of something" (1). In the past few years, when we talk about chemical visualization, we typically mean computer models. Although this type of visualization is extremely important, we must remember that different people visualize with different models -- just as we accept the realization that different people learn in different ways (Multiple Intelligences). BODY OF THE TEXT: One of the main global problems that still exists in attracting students to study chemistry is that many teachers still do not take into account the different styles of learning which different students have, such as the "Multiple Intelligences," by Howard Gardner of Harvard University (2). Lately,
"visualization" has become a buzzword, in both science
and education. The
word "visualize" is defined as "To form a mental
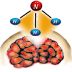
image or vision of; picture in the mind". Joe Nelson, a student
at Columbia College, created the following artwork to illustrate
the dictionary definition of visualization, as well as the process
of visualizing a chemical bond in the mind:
The
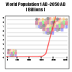
world's population is growing exponentially, and in October 2000
it has passed the 6,000,000,000 mark - as can be seen in the following
graph: We
must develop methods to make science accessible to all the world
population in order to avoid a class society divided -- not by royalty
-- but by knowledge of science. In
order to avoid getting into this situation, we developed methods
of teaching science that incorporate art, music, dance, drama, and
cultural backgrounds (6). In the Science Institute at Columbia College,
we do not practice what is termed "chalk and talk" teaching.
A
group of students majoring in theater joined together to write and
act out an original dramatic presentation of "Sodium and Chlorine:
A Love Story", presented as a mock Shakespearean tragedy, for
their chemistry class project. One of these students has become
a famous actor; when the author went to see him backstage at a performance,this
former student stated that he had forgotten many things he learned
in school, but he vividly recalled the ionic bond, the Periodic
Table, and other concepts from his chemistry class almost fifteen
years earlier, because he had used theater to visualize these concepts.
Another
Columbia student created an entertaining multimedia project titled
"The Amazing Atom" to visualize the ionic bond of sodium
and chlorine, and to illustrate different models of the atom.
This
method has also been employed in workshops conducted by the Science
Institute to enhance the teaching skills of Chicago Public School
teachers. These teachers create their own projects to communicate
their understanding of scientific concepts covered in the workshops,
as well as practice this method in their own classrooms. A group
of teachers created "The Element Connection" as a parody
of "The Love Connection" television show. The
effectiveness of employing these methods in Chicago Public School
classes can be seen by the excellent (and scientifically accurate)
projects created by children, such as this artwork made by a seventh
grade student: Chicago
Public Schools teachers who participate in Science Institute workshops
and employ these methods in their classes have reported that seventh
and eighth grade children (who previously would "never come
close to the chemistry lab") now prefer to stay after school
in chemistry clubs, rather than attending gym class. Many of these
students have chosen to attend high schools which specialize in
science and mathematics -- which never happened before in these
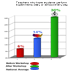
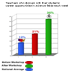
teachers' experience. These graphs show the remarkable increase
of teacher's classroom practices after the workshops in a (a) conducting
science experiment daily or almost every day and (b) discussing
with their students science careers weekly, compared to practices
before the workshop and compared to national averages.
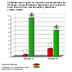
These
methods have proven particularly effective at what our experience
demonstrates are the crucial ages: the fifth grade (before children
enter middle school), and the eighth grade (before children enter
high school). This graph represents the achievement of students
of teachers who attended our workshops, compared to those of students
in the same school whose teachers did not attend our workshops.
ACKNOWLEDGEMENTS
BIBLIOGRAPHY
Last updated 15.05.02 |
|
| CEI is a newsletter of the Committee on Chemistry Education (CCE) of IUPAC |
 |
 |
 |
 |