|
 |
 |
 |
|
Chemical Education International, Vol. 3, No. 1, AN-1, Received October 25, 2001
J.
J. Lagowski INTRODUCTIONIn
1997, on the occasion of a visit by A. F. M. Fahmy to The University
of Texas at Austin, extensive discussions between A. F. M. Fahmy
and J. J. Lagowski laid the basis for the development of the Systemic
Approach to Teaching and Learning (SATL) Chemistry. Our interest
in developing the SATL strategy arose from the recognition of the
increasing globalization of a wide spectrum of human activities
such as economics, media, politics, and entertainment. We are not
interested here in a value judgment concerning globalization, rather
our position is that globalization is occurring at a rapid rate
and we have an obligation to our students to help them cope with
this phenomenon. Globalization implies a broader context when considering
human activities than a regional or a local perspective. Specifically,
our interest is in a vision of science education--that process by
which progress in science is transmitted to the appropriate cohort
of world citizens--that is sufficiently flexible to adapt to an
uncertain or, at best, an ill-defined future. That future, however,
ultimately must include an appreciation of the vital role that scientists
and chemists, in particular, have in human development. Thus, the
future of science education must reflect a flexibility to adapt
to rapidly changing world needs. It is our thesis that a systemic
view of science with regard to principles and their internal (to
science) interactions as well as the interactions with human needs
will best serve the future world society. Through the use of a systemic
approach, we believe it is possible to teach people in all areas
of human activity--economic, political, scientific, as well as ordinary
citizens--to exhibit a more global view of the core science relationships
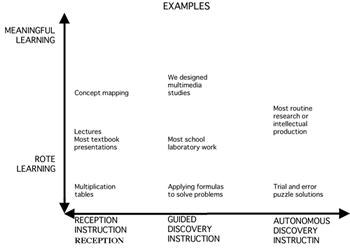
and of the importance of science to such activities. As a start, we suggest the development of an educational process based on the application of "systemics" (vide infra), which we believe, will affect both teaching and learning. The use of systemics, in our view, will help students begin to understand interrelationships of concepts in a greater context, a point of view that ultimately should prove beneficial to the future citizens of a world that is becoming increasingly globalized. Moreover, if students learn systemics in the context of learning chemistry, we believe they will doubly benefit by learning chemistry and learning to see all subjects in a greater context. Concept Mapping and Systemics. In retrospect, the key feature of systemics (SATL) can be imagined as an extension of concept mapping. In the early 1960's, when behaviorist theory prevailed among educational psychologists, Ausubel published his theory of meaningful learning, portions of which appeared in his book (1963) entitled "The Psychology of Meaningful Verbal Learning" (1); a more comprehensive view of his ideas was published later (2). Contemporary assimilation theory stems from Ausubel's views of human learning which incorporates cognitive, affective, and psychomotor elements integrated to produce meaningful learning (as opposed to rote learning). To Ausubel, meaningful learning is a process in which new information is related to an existing relevant aspect of an individual's knowledge structure and which, correspondingly, must be the result of an overt action by the learner. Teachers can encourage this choice by using tools such as concept maps (3). It is postulated that continued learning of new information relevant to information already understood (presumably) produces constructive changes in neural cells that already are involved in the storage of the associated knowledge units. In our view, an important component in Ausubel's writing has been the distinction he emphasized between the rote-meaningful learning continuum and the reception-discovery continuum for instruction. The orthogonal relationship between these two continua is illustrated in Figure 1.
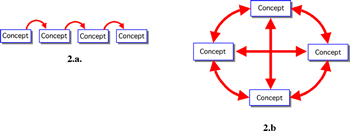
According to Ausubel, the essence of the meaningful learning process is that symbolically expressed ideas are related to what the learner already knows. Thus, in a sense, Ausubel's work also incorporates the elements of contructivism. Meaningful learning presupposes that the learner has a disposition to relate the new materials to his or her cognitive structure and that the new material learned will be potentially meaningful to him or her. In other words, it takes an overt act by the learner to make learning meaningful. Concept mapping (3) is a device that can be used to communicate to the learner as well as providing a vehicle to help the learner with meaningful learning tasks (vide infra). Of the types of meaningful learning that Ausubel described--representational learning, propositional learning and concept learning--the latter is of interest here. The acquisition of subject matter primarily consists of concept learning. Concept mapping (3) is a device that provides the basis of relating new knowledge to previously assimilated knowledge in a systematic way. Concept mapping also incorporates a strong element of constructivism, in the sense that a student can build his/her understanding of new concepts on those he/she has a deep familiarity. Concept maps have been used as metacognitive tools to help teachers and learners to improve both teaching and learning. Concept maps created by students are an idiosyncratic representation of a domain specific knowledge and provide teachers with information on what students know because such maps can show the students initial concepts, how they are contextually related, and how learners reorganize their cognitive structures after a special teaching activity. It might be tempting to try to use concept maps as the focus for the assessment of students' acquisition of concepts, however, a number of observations on attempts to "score" concept maps suggest that, currently, they are not good assessment tools (4), mostly because there are many possible concept maps that "correctly" show the relationships among a given collection of concepts. THE SYSTEMIC APPROACH TO TEACHING AND LEARNINGWe introduce now the basic ideas of the systemic approach to teaching and learning (SATL) as applied to chemistry. By "systemic" we mean an arrangement of concepts or issues through interacting systems in which all relationships between concepts and issues are made clear, up front, to the learner using a concept map-like representation. In contrast with the usual strategy (3) of concept mapping, which involves establishing a hierarchy of concepts, our approach strives to create a more-or-less "closed system of concepts"--a concept cluster--(Figure 2b) which stresses the interrelationships among concepts; Figure 2 also illustrates diagrammatically the difference between a linear representation of concepts (2a) and our systemic representation (2b).
We
believe it is more difficult to obtain a global view of a collection
of linearly arranged concepts (2a) than with the systemic representation
(2b), which stresses all relationships among concepts. Further,
our use of the term "systemics" stresses recognition of
the system of concepts that form the closed cluster of concepts
under consideration. "Systemics" means in our hands, the
creation of closed-cluster concept maps for the purposes of helping
students learn; "systemics" is an instructor-oriented
tool and, hence, requires teacher and student materials to be created
about the closed-cluster concept map strategy.
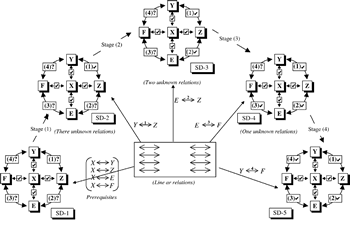
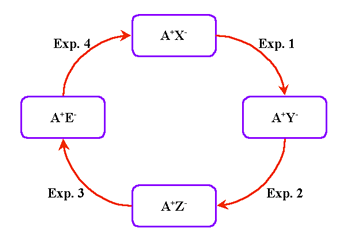
The instructor has in mind the concept structure shown in Figure 2a, which he/she wants to develop into the closed cluster shown as Figure 2b. The prerequisites are simple bi-directional relationships between the concepts. Thus, initially, there are four unknown (to the student) relationships in the final cluster of concepts (Figure 3). The full closed cluster concept map can be developed in four stages by sequentially introducing the (initially) four unknown concepts. At each step, another part of the final closed concept cluster is added and developed. This process clearly illustrates the systemic constructivistic nature of our SATL approach. THE APPLICATION OF SYSTEMICS TO CHEMISTRY INSTRUCTIONAfter a preliminary study of the application of SATL techniques in secondary schools in Cairo and in Giza, we embarked on a number of studies to probe the extent (content material-subject matter) and level of instruction that could be effectively expressed using such methods and whether such methods helped students learn. A list of these studies is given in Table I. All of these studies required the creation of new student learning materials (as shown in Table 1), as well as the corresponding teacher-oriented materials. Table
1. A list of experiments conducted using the SATL strategy using
various aspects of the subject chemistry. The initial experiment
was conducted in 1998 in the secondary schools of the Cairo and
Giza school districts.
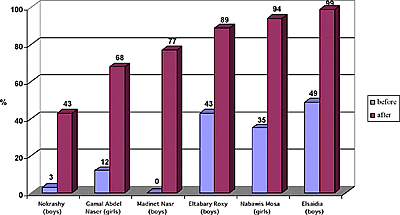
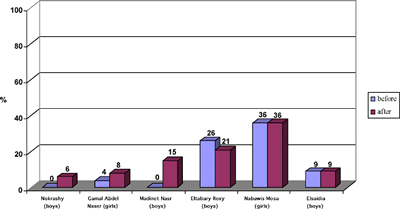
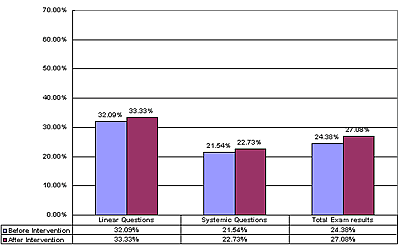
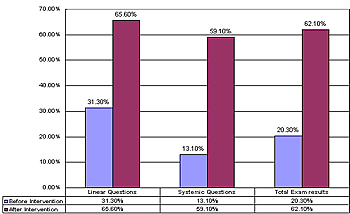
The statistical analysis of student achievement results shows that the students engaged with SATL materials and taught by teachers trained in systemics achieve at significantly higher levels than those taught by the standard linear methods. More SATL chemistry courses were produced by the Science Education Center at Ain Shams University, which are still under experimentation in different university settings (e.g., Inorganic Chemistry and Systemic Organic Experiments). A description of these courses was presented at the 1st Arab Conference on Systemic Approach in Teaching and Learning, Organized by the Science Education Center at Ain Shams University and the UNESCO regional office in Cairo, Egypt in February 2001. SATL COURSE EVALUATIONWhen it is feasible, we attempt to evaluate the SATL courses that have been created using student achievement as primary criterion for success. Our evaluation strategy generally involves experimental groups of students that use SATL materials taught by instructors trained in SATL methods (Figure 2b) and an equivalent (as far as background is concerned) control group of students taught by conventional methods, which are often based on a linear strategy (Figure 2a). Pre-college courses. Our initial experiment probing the usefulness of the SATL approach to learning chemistry was conducted at the pre-college level in the Cairo and Giza school districts. Nine SATL-based lessons in organic chemistry (9) taught over a two-week period were presented to a total of 270 students in the Cairo and Giza school districts; the achievement of these students was then compared with that of 159 students taught the same material using standard (linear) methods. The details of a statistical summary of the results of achievement tests on these experimental groups before and after the SATL treatment and for the reference group before and after the same material was presented in the conventional linear way is available for inspection (5), but we address here only the overall results (Figures 4 and 5) for the sake of brevity. The results indicate that a greater fraction of students exposed to the systemic techniques, the experimental group, achieved at a higher level than did the control group (taught by conventional linear techniques). These results are statistically significant (5). The experimental (SATL) (Figure 4) and control group (Figure 5) are separated into the individual schools in the Cairo and Giza school districts.
All of the students were tested before the experiment began to establish the common background of these groups. The experimental group was taught by SATL-trained teachers using SATL techniques with specially created SATL materials, while the control group was taught using the conventional (linear) approach. Our results from this pre-university experiment point to a number of conclusions that stem from the qualitative data (5), from surveys of teachers and students, and from anecdotal evidence.
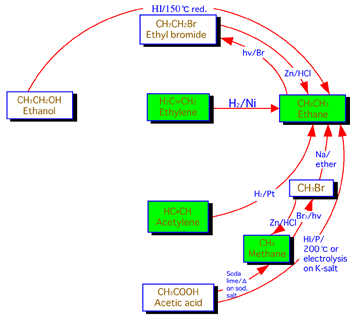
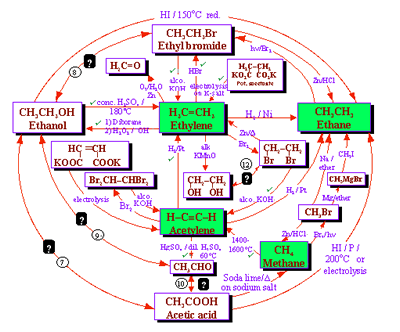
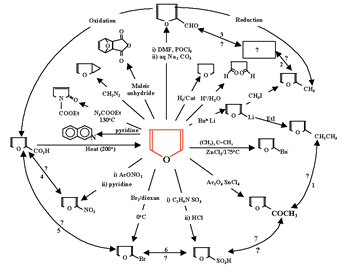
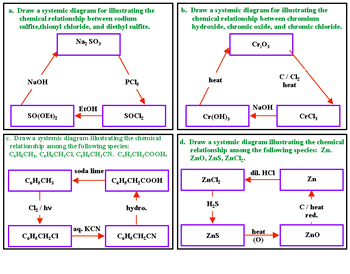
Organic chemistry: A study of the efficacy of systemic methods applied to the first semester of a typical second year organic chemistry course (16 lectures, 32 hours) at Zagazeg University was conducted after the usual course materials on aliphatic chemistry were converted to a systemic approach (6). We present now the details of the transformation of the usual linear approach usually used to teach this subject that involves separate chemical relationships between alkanes and other related compounds (Figure 6) and the corresponding systemic closed concept cluster that represents the systemic approach (Figure 7).
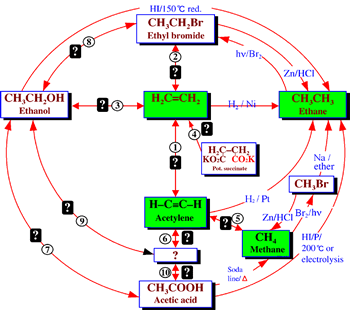
This systemic diagram in Figure 7 can be constructed from Figure 6 by answering the following questions:
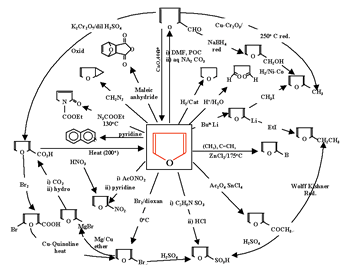
The answers to these questions are displayed in the systemic diagram shown in Figure 7. Notice that, in Figure 7, some chemical relationships are defined whereas others are undefined. The undefined chemical relationships are collected in Table 2. These undefined relationships are developed systematically. After a study of the synthesis and reactions of alkenes we can modify the systemic diagram shown as Figure 7 to accommodate other chemistries as shown in Figure 8.
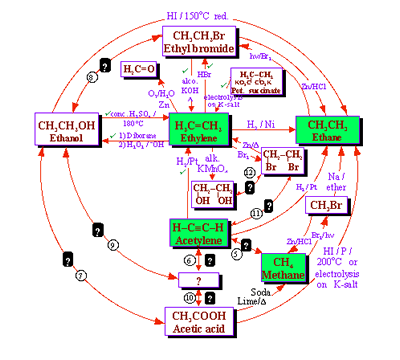
Expanding the chemistry of acetylene converts the systemic diagram in Figure 7 to that shown in Figure 9.
In
a similar fashion, the systemic diagram shown in Figure 7 can accommodate
to the chemistries of ethyl bromide and ethanol yielding a new systemic
diagram.
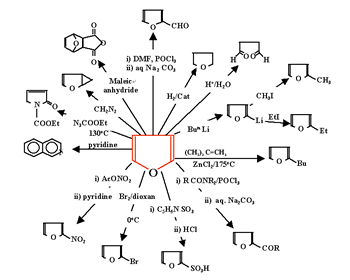
Heterocyclic chemistry: A course on heterocyclic chemistry using the SATL technique was organized and taught to 3rd year students at Ain Shams University. A portion of the one-semester course (10 lectures, 20 hours) was taught to 53 students during the 1999-2000 academic year. We use heterocyclic chemistry to illustrate, again, how a subject can be organized systemically. Figure 12 summarizes all the significant reactions of furan, the model heterocyclic compound.
These
are the reactions that are generally discussed in a linear fashion
(Figure 2a) in the conventional teaching approach. However, these
reactions can be organized systemically as shown in Figure 13.
But inspection of Figure 13 reveals seven unknown relations among the furan compounds; these are listed in Table 3.
Figure 13 can be further refined to give Figure 14 by adding the unknown chemical relations shown in Table 3.
Before the intervention, that is, teaching by systemics, the students in this class all took an examination--we call the "zero point" examination--which established their previous personal knowledge of organic chemistry as obtained from previous courses. The data summarized in Table 4 show that students taught systematically improved their scores significantly after being taught using SATL techniques.
These results are statistically significant at the 0.01 level. Statistical details are available from AFMF (9). SYSTEMICS AND LABORATORY INSTRUCTIONIn this section, we discuss the use of systemics in laboratory instruction, specifically, conducting experiments that reveal the "chemistry of species" of interest (10). Applying Systemics to this phase of laboratory instruction reveals the following advantages:
We start our discussions with the classical laboratory-oriented subject of qualitative analysis, which involves collecting chemical information on the species of interest from which pool of information the identities of the species of interest are deduced. Qualitative analysis involves the application of linearly obtained chemical information to an unknown solution in a linear way (Figure 15).
In contrast to the generally linear approach of learning descriptive chemistry of cations from a laboratory experience, a systemic method has been developed that focuses attention on individual species (Figure 16).
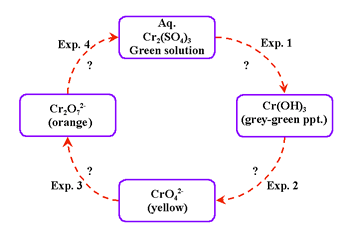
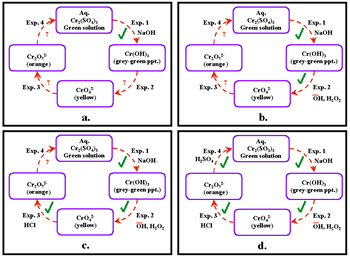
The reactions can be performed in a single test tube on a small sample (=<0.5ml). This approach allows students to experience the colors of chemical species, their solubility characteristics, and their redox behavior. The "green chemistry" aspects of this approach involve a very small amount of the cation-containing species, which is contained in a very small volume. We now show the development of a systemic closed-cluster cycle (Figure 17). This diagram shows the qualitative investigation of Cr3+ species, the preparation of Cr3+ compounds, and the interconversion of the species.
Notice that the formulas of chemical species of interest are expressed in this diagram, but the reagents that bring about these conversions are not given in the diagram in Figure 17. These reagents are revealed experimentally in a series of reactions shown in Figure 18a-d, which the students can do in the laboratory on a small single sample of chromium-containing species.
Thus, the experiment diagrammed in Figure 18a reveals that hydroxide ion (OH-) converts the green aqueous solution of Cr2(SO4)3 into the grey-green precipitate of Cr(OH)3; the addition of hydrogen peroxide converts the green precipitate into the soluble, yellow-colored chromate ions (CrO42-); and so forth. The systemic technique can also be used to create an activity that develops equation-writing skills (Figure 19).
We suggest that the systemic diagrams shown in Figure 19 can be used as a new assessment tool in chemistry (Systemic Assessment). TRAINING PROGRAMSOver
and above the activities involving the creation and evaluation of
systemic materials, the Science Education Center (SEC) has been
engaged in the last two years in training programs using the systemic
approach. These programs included Chemistry Professors from several
Egyptian Universities where the training is focused on using the
systemic approach in teaching chemistry at the university level.
In addition, training programs were carried out for pre-college
teachers, which involved video conferences attended by about 8,000
school teachers as well as smaller groups in face-to-face training
sessions. SUMMARYWe have attempted to describe the application of systemics to a variety of chemistry-oriented courses starting with pre-college instruction through advanced organic chemistry subjects as well as laboratory-oriented instruction. In addition, we have presented the results of the evaluation of this approach on student performance. Literature Cited(1) Ausubel, D. P., The Psychology of Meaningful Verbal Learning; Grune & Stratton: New York, 1963. (2) Ausubel, D. P., Educational Psychology: A Cognitive View; Holt, Reinhard and Winston: New York, 1968. (3)
[a] Novak, J. D. and Gowin, D. B., Learning How to Learn;
Cambridge University Press: Cambridge, 1984. (4) Stuart, H. A., Eur. J. Sci. Educ., 1985, 7, 73. (5) Fahmy, A.F.M., Lagowski, J.J.; Systemic Approach in Teaching and Learning Carboxylic Acids and Their derivatives, http://www.salty2k.com/satlc.html (6) Fahmy, A.F.M., Lagowski, J.J.; "Systemic Approach in Teaching and Learning Aliphatic Chemistry"; Modern Arab Establishment for printing, publishing; Cairo, Egypt (2000). (7) Fahmy A. F. M., El-Hashash M., "Systemic Approach in Teaching and Learning Heterocyclic Chemistry". Science Education Center, Cairo, Egypt (1999). (8) Fahmy A. F. M., Hashem, A. I., and Kandil, N. G.; Systemic Approach in Teaching and Learning Aromatic Chemistry. Science, Education Center, Cairo, Egypt (2000). (9) Fahmy A. F. M., SATLC in Egypt; report to CTC. Meeting, Brisbane, Australia (July 2001). (10) Fahmy, A. F. M.; Hamza M. S. A; Medien, H. A. A.; Hanna, W. G.; and Lagowski; J. J.; Satellite conference of the WCC 2001; Brisbane, Australia 1st July (2001): http://www.salty2k.com/satlc
Last modified 21.05.02 |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CEI is a newsletter of the Committee on Chemistry Education (CCE) of IUPAC |
 |
 |
 |
 |