|
 |
 |
 |
|
Chemical Education International, Vol. 3, No. 1, AN-2, Received in February 24 , 2002
Primary and Secondary Science Education in Japan at a Crisis Point TAKEUCHI
Yoshito Abstract: Primary and secondary school science education in Japan, which once had a reputation that was recognized worldwide, has lost its past excellence due primarily to continual reductions in teaching material content and school hours. The positive and negative role of the course of study (COS) and the school textbook screening system (STSS) is discussed, and steps are proposed to improve the situation. 1. High Estimation of Science Education in Japan The author has been involved in primary and secondary science education in Japan in various ways: as the editor-in-chief of science textbooks prescribed for primary and junior high school education, and of chemistry textbooks for senior high school. Another of his activities has been the promotion of chemical education in Japan to the world. As a part of activities of this, Prof. M. M. Ito and the author of this paper published two English-language books, "Chemical Education in Japan, 2nd version"2 and "Chemical Education in Asia/Pacific"3 from the Chemical Society of Japan (CSJ). The latter publication was a joint project between the CSJ and the Federation of Asian Chemical Societies (FACS). Fifteen chemical societies belonging to FACS contributed a chapter to the publication "Chemical Education in Asia/Pacific" to outline the fact and figures of their chemical education policies. Through these kinds of activities we received much praise to the effect that the remarkable economic development of Japan after the World War II was due very much to its success in primary and secondary science education. We are of the opinion that Japan deserved such praise in the past; however in the present situation and most likely in the future, such praise is not warranted. Rather, we fear that primary and secondary science education in Japan is at a crisis point. For these reasons the author wishes to contribute this report to science education authorities around the world. It is generally accepted that the success of Japanese science education in the past was the results of many factors such as:
2. The Course of Study as the Guideline of Science Education in Japan It is generally well known that the Japanese Government controls primary and secondary education by two systems; the course of study (COS) and the school textbook screening system (STSS) strictly control the content of primary and secondary science education in Japan. Before World War II, school textbooks were compiled by the state. COS could be regarded as the result of postwar democracy, with its original purpose being to provide a kind of loose set of guidelines for the authors and publishers of textbooks. The idea was gradually changed into a strict set of regulations such that now, on the basis of STSS, no authors or publishers can go against Government recommendations concerning textbooks.This has become a serious problem in terms of authors wishing to publish good textbooks that may be stimulating to pupil/students due to the introduction materials outside the scope of COS. COS is generally revised every ten years. Thus, as the time of revision of COS approaches, the Government will organize several review committees, such as the Central Education Council (CEC) for example. CEC usually advocates a new doctrine (Table 1) for the coming decade, and in addition will suggest the addition and/or deletion of teaching material from COS. The setting of the number of school hours is also within its jurisdiction. Table 1 Doctrines Featured in COS
The 1st COS featured "science based on everyday life", while the 2nd COS focused on Òsystematic study of scienceÓ as is indicated in Table 1. During the first two COS periods school hours remained essentially unchanged or even increased to some extent. It must be noted that COS played a positive role in primary and secondary education in Japan in the past as a means to maintaining the high standard of the primary and secondary science education. The problem however, arose with the COS of 1977.
3. COS of 1968: the Sputnik COS. On October 4, 1957, the USSR successfully launched the first spacecraft Sputnik 1. Shocked by the great success of a rival country, the US Government decided to reform its science education policy in a relatively rapid and dramatic way. This was in response to the view that the reason why the USA was beaten in the space development program was because of inadequacies in its science education system. A massive commitment of money and manpower was immediately carried out, with the result of this effort being a series of excellent curricula and textbooks for senior high school science. As for chemistry, "CBA (Chemical Bond Approach) 4" and "CHEMS (CHemical Education Material Study) 5" should be mentioned. For physics and biology, similar curricula and textbooks were also prepared. The basic idea of these new curricula, CBA in particular, was the modernization of science education by stressing theoretical aspects of science much more than what appeared in existing curricula and textbooks. In CBA, the nature of chemical bonding was thoroughly described by introducing such advanced concepts as the orbital and its energy levels, and hybridization. To understand the thermodynamic aspects of chemical reactions more theoretically, the concept of entropy was introduced in a qualitative way. These curricula and textbooks were issued in the early 1960s, and naturally they had a powerful effect on science education in other countries, particularly on that in Japan. The COS of 1968 can be regarded as a product of innovations to science education made by the Americans. For this reason we have chosen to call this COS the "Sputnik COS". Such concepts as orbital, hybridization and entropy were also included in the Sputnik COS. The process of modernization was probably much too rapid for science education in Japan as well as for that in the USA. Many students were unable to follow the lessons. It is said that there were some teachers who had difficulty in understanding the concepts introduced in new textbooks. There must have been much criticism of this rather impetuous wave of innovation. As a result, the subsequent COS of 1977 saw a nearly complete withdrawal from the Sputnik innovation. The doctrine of this latter COS was Òconcentration on fundamental items".
4. Serious Reduction of Course Content and School Hours The most serious problem that started from the COS of 1977 was a CONSTANT reduction of COS content and of hours of school attendance in primary and secondary science education. The undigested attempt of innovation has to be one of the reasons for this rapid retreat. There is also another reason: with a continuously increasing number of children other than just the brightest children attending senior high school each year, the average capability of students must by definition become lower than before. The situation has to be much the same in the USA. The
feature of the COS of 1977 and subsequent ones may be summarized
as a continuous reduction of course content and of school hours.
When the new COS comes into being in a few years, the amount
of course content and of school hours will be probably 2/3 or
even less as compared with maximum values from previous versions
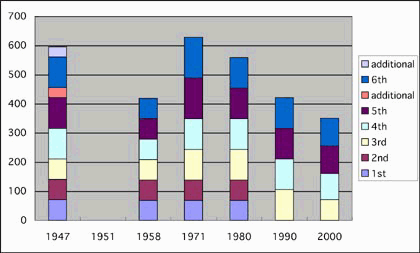
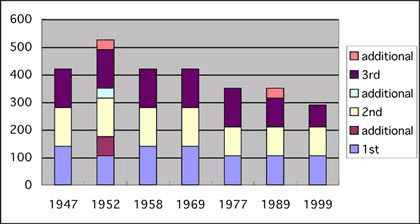
of COS. In Figs 1 and 2, increases and decreases of school hours
for science lessons in primary and secondary schools are summarized.
The particularly large decrease of school hour for primary school that began with the COS of 1977 was a direct outcome of the complete removal of science lessons from 1st and 2nd year curricula. The Government introduced a new subject, "life environment studies", which is something that is far removed from science and more related to social morality training.
5. The New Course of Study; the Crisis of Primary and Secondary Science Education in Japan The new COS, which will be effective from 2002 for primary and junior high schools and from 2003 for senior high schools, clearly demonstrates this tendency of uniform reduction of course content and school hours. Two examples from the field of chemistry bear witness to the significance of the reduction and problems caused by this. Previously,
in junior high school, ions and related topics such as electrolysis
were taught immediately after atoms and molecules had been taught,
which meant that pupils were provided with a general idea of
the three fundamental constituents of matter: atoms, molecules
and ions. What would happen if the concept of ions were to be
deleted from the textbook? It is impossible for teachers to
teach the chemistry of NaCl in an appropriate manner. It should
be appreciated that electrolysis has served as one of the most
attractive subjects in elementary chemistry since the time of
Michael Faraday (Fig. 3). What children of the early 19th century
could enjoy is now beyond the reach of children of the 21st
century.
It must be added that CSJ was not necessarily without resistance. Whenever the Committees of the Government announced their reports6 describing the policy of the new COS (substantial reduction of course content and school hours), CSJ issued critical comments, sometimes independently and sometimes with other scientific societies. All these efforts were in vain. The second example is from the chemistry curriculum for senior high school. Previously, and naturally, students would learn about chemical bonds immediately after they had studied atoms, molecules and ions. In the new COS, atoms, molecules and ions are separated from chemical bonds due to the formation of two different subjects, Chemistry I and Chemistry II (Table 2). Table 2. Structure of Chemistry Course in the New COS Chemistry I
Chemistry II
One may argue that this separation would not be such a problem since students will eventually encounter and learn chemical bonds in Chemistry II. This is not, however, true. It is likely that most students who would choose science and technology as their future occupation will study Chemistry I, but not all of them will study Chemistry II. In fact, it is predicted that less than half of Chemistry I students would be likely to study Chemistry II. This is the outcome of the doctrine of the COS of 1989 in which it was advocated that more attention should be paid to individual students. What this actually meant was that there should be more optional subjects so that individual students could select subjects according to their future plans. The principle sounds reasonable, but the reality is not so clear-cut. The trouble is that with the decreasing number of compulsory subjects, there was now simply no need for most students to choose Chemistry II in addition to Chemistry I. The number of required subjects for qualification was so small that it would be enough for students to choose Chemistry I plus one other simpler subject. The results of such education policy decision making will now become clear. There will be many senior high school graduates who will finish studying chemistry without learning about chemical bonds. The reason should now be clear why we stress that primary and secondary science education in Japan is at crisis point. In other words, primary and secondary science education in Japan has undergone a rapid and continuous deterioration since the COS of 1977.
6. Factors Accelerating This Tendency It should be pointed out, however, that there are other factors responsible for the deterioration described above. The most problematic factor is the university entrance examination, which in fact determines the mode of senior high school education. Japan is now a country with fewer children than 10 years ago, and so the number of children who would normally have gone to universities in that time has also decreased. However, the full complement of universities has remained essentially unchanged. This situation has caused intense competition among universities to recruit freshmen/women. The simplest and probably most effective means to attract candidates is to reduce the number of subjects for the entrance examination. This certainly affects the line of thinking of the senior high school students. Most of them might feel that it is not necessary, or perhaps even absurd, to select subjects that will not be required at the entrance examination for the university that he/she would like to enter. Even today, most university lecturers find difficulty when they first teach freshmen/women because the backgrounds of these students are so diverse. There are some freshmen/women in Department of Chemistry who did not choose chemistry as the subject to be examined at the entrance examination. In such cases their background in chemistry is usually poor even if they studied chemistry at senior high school. It seems that for senior high school students it is difficult to find a motivation for studying any subject that is not explicitly related to the university entrance examination. The university entrance examination under the new system will start from 2006, and hence it would be premature for one to estimate how many universities will require candidates for science and technology departments to assign both Chemistry I and II at the entrance examination. The author of this paper fears that from 2006 there could be many freshmen/women in the faculty of science, or even in the department of chemistry who have studied chemistry but did not study anything about chemical bonds at the senior high school level.
7. How to Reactivate Primary and Secondary Science Education in Japan Is it then possible to reactivate primary and secondary science education in Japan? In view of the analyses described above, effective solutions may be necessarily rather drastic. One measure is the abolishment of COS. The author of this paper admits the positive role of COS in the past. The reason why COS lost its initial mission is that COS adheres to a policy of reducing the maximum amount of content in COS. If the role of COS is to delineate minimum requirements for students, then its role will be more positive. The second measure is the abolishment of STSS. The author believes, based on his experiences as the editor-in-chief of textbooks which suffered under STSS, that STSS carries more responsibility than COS for the deterioration of primary and secondary science education in Japan. COS in effect provides a set of guidelines (of maximum content) which the authors of textbooks must follow. In order to encourage better students/pupils, authors might try to conceal some hints by which teachers could introduce some topics beyond those set down by COS. Such attempts have so far been ordered to be deleted from the textbooks by Government textbook inspectors. Consequently, though several publishers might publish textbooks on a particular subject, it is very difficult to find any significant difference between the textbooks. The opportunity for devoted teachers and absorptive students to extend their interests to advanced topics is therefore lost. The role of STSS should therefore be restricted to an investigation of mistakes and errors in the textbooks. According to a recent newspaper account, the Government intends changing its policy concerning STSS, with more freedom being given to authors/publishers. The author of this paper, however, is not so optimistic as to believe that the Government will change its long lasting policy. For this reason this author will continue to announce to the world the fact that primary and secondary science education in Japan is at a crisis point. Keyword: course of study (COS); school textbook screening system (STSS); CBA (chemical bond approach); CHEMS (chemical education material study). References
Posted April 20, 2002.
Last modified 19.07.02 |
|
| CEI is a newsletter of the Committee on Chemistry Education (CCE) of IUPAC |
 |
 |
 |
 |