|
|
Vol.
34 No. 1
January-February 2012
by Gavin O’Connor and Louise Dean
Measurement affects almost every aspect of our daily lives, from the fuel in our cars and the quality of the air we breathe, to the safety of our food, water, healthcare, and our cosmetic products. Chemical and bioanalytical measurements in particular underpin the enforcement of regulations. Every day, thousands of chemical measurements support decisions on food safety, health, and environmental protection. It is therefore critical that organizations producing or using measurement data have confidence in the results obtained.
Today’s global economy highlights more than ever the need, both nationally and internationally, for confidence in measurement results. A key step in achieving this is to ensure that results can be related to suitable, internationally recognized references. “Metrological traceability”1 is the property of a measurement result whereby the result can be related to a recognized reference through a documented unbroken chain of calibrations, each contributing to the measurement uncertainty.2 To enable comparison of measurement results over time and distance it is often beneficial for the reference to be a base unit of the International System of Units (SI). The availability of suitable measurement standards for calibration is fundamental to ensuring traceable results.
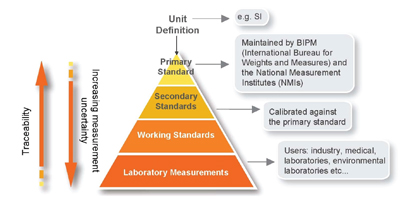 |
| Figure 1: Traceability Chain Linking a Laboratory Result to the Definition of a Measurement Unit. |
The international infrastructure for measurement was initially developed for physical measurements of quantities such as mass and length. Primary international and national measurement standards exist which are used as the references on which to base the traceability of measurement results. We take for granted that items can be weighed accurately, or that we can find out the time anywhere in the world. In the physical model, traceability is achieved by comparisons or calibrations which lead directly to a defined unit, for example, the international prototype kilogram. Figure 1 shows a schematic of a “traceability chain” linking a laboratory result to the definition of a measurement unit. A similar infrastructure has been developed for chemical measurements, but the situation is often more complex than that for physical measurements.
Chemical measurements encompass a vast number of elements and compounds in a diverse range of sample types. There is therefore a requirement for a wide range of different measurement standards2 to be able to establish the traceability of chemical measurements. In addition, to be able to measure the amount of a particular chemical entity, test samples often require significant pre-treatment to separate the analyte(s) of interest from the sample matrix. Measurement procedures used in chemical analysis are therefore frequently multistage, each measurement step potentially introducing factors which could influence the result. Chemical measurements may therefore involve a number of measurement standards for physical quantities such as mass and volume, and certified reference materials for establishing the traceability of amount of substance.
A certified reference material is a material of known homogeneity and stability and is accompanied by documentation providing one or more specified property values with associated uncertainties which have been obtained using reference measurement procedures.2 In chemistry, these materials are typically classified as either pure substances (which can be used for the preparation of calibration solutions) or matrix materials (used, for example, to evaluate measurement bias as part of method validation). It is imperative that laboratories have access to well characterized matrix reference materials that are a good match to real test samples. One of the reasons why such materials are so sought after for chemical measurements is that the sample matrix often has a significant effect on method performance.
The National Measurement Institutes (NMIs) around the world develop and maintain national measurement standards. LGC, the UK’s designated NMI for chemical and bioanalytical measurement, supports the measurement infrastructure in the UK by producing reference materials that enable other laboratories to ensure the traceability of their chemical and biological measurement results through instrument calibration and method validation. At the heart of the measurement infrastructure is the ability of the NMIs to characterize the amount of substance for well-defined measurands2 (analytes) using reference measurement procedures that can provide measurement results deemed fit for purpose with a direct and unbroken link to a base unit of the SI.
Exact Matching Isotope Dilution Mass Spectrometry, a “higher order” reference measurement procedure, is an iterative process for providing measurement results with an unbroken traceability chain to the SI. Here, certified reference materials go some way to fulfilling the role of primary standards in the metrological framework. The huge variation in potential matrices and measurand concentration levels mean that there is no shortage of need for new reference materials.
Although traceability in chemical and physical measurement is well established, the biological measurement field is at a much earlier stage in its development, and clinical measurements in particular present their own set of unique problems. Poor-quality clinical measurements can lead to misdiagnosis or incorrect prescription of medicine for patients. It is therefore essential that certified reference materials are available for manufacturers to establish the traceability of values assigned to calibrators supplied with diagnostic equipment, and for medical and clinical laboratories to validate their methods. The In Vitro Diagnostic Medical Devices Directive (EC IVDD, 98/79/EC) stipulates “the traceability of values assigned to calibrators and/or control materials must be assured through available reference measurement procedures and/or available reference materials of a higher order.” This ensures harmonization of standards across Europe, which in turn provides a high level of health protection for patients. It is therefore necessary that high-accuracy, low-uncertainty reference materials are produced to satisfy the objectives of the directive. At present, the number of IVD devices and potential measurands vastly exceeds the number of higher-order reference materials available. The NMIs are now addressing this problem by developing such materials to help IVDD compliance. However, it is not unusual for different analytical platforms, calibrated using different reference materials, to be used for the same measurand. There is therefore a need across the clinical community for commutable reference materials that ensure traceable measurement results, independent of the analytical platform used.
The following examples demonstrate how LGC is supporting the clinical sector in ensuring sound measurement practice to underpin clinical efficacy, quality assurance, and patient safety.
Immunoassays and spectroscopic or enzymatic techniques are highly desirable for high-throughput screening methods due to the high speed and relative low cost of the technology. However, it has been well documented that these routine methods can suffer from interferences that result in measurement bias. In order to reduce the possibility of poor-quality clinical measurements, and to assist manufacturers in satisfying the objectives of the IVDD, LGC has developed a primary ratio measurement procedure,2 exact matching IDMS, for assigning reference values to a number of certified reference materials for routine health screening. The reference materials, aimed at the clinical sector, cover steroids and therapeutic drugs.
The production of certified reference materials for a therapeutic drug poses different challenges. Many therapeutic drugs have a narrow therapeutic range. The dose given to patients is often regulated by the amount of drug substance circulating in the blood. Therefore, accurate measurement has a direct impact on clinical intervention.
Tacrolimus is an immunosuppressant drug given to patients after an organ transplant. Once an organ is transplanted, a complex therapeutic regimen is undertaken to prevent rejection of the allograft and to allow a foreign body to remain functional within the recipient. Due to the narrow therapeutic range of tacrolimus, routine therapeutic drug monitoring of a patient’s blood is essential to prevent acute rejection and ensure long-term survival of both the patient and the allograft.
Tacrolimus is measured by a variety of liquid chromatography mass-spectrometric (LC-MS) and immunoassay-based methods, which are all independently calibrated, but without agreement to a common reference or to an accepted reference method or standard tacrolimus reference material. This means that mass concentration values may not be comparable between methods or laboratories, posing potential risks to patients undergoing therapeutic drug monitoring. In severe cases this can lead to the patient either receiving an insufficient dosage and rejecting the organ, or receiving a high, toxic dose.
This lack of traceability has been identified by medical laboratories and diagnostic kit manufacturers as an issue when trying to achieve consistent measurement comparability and in setting reliable therapeutic levels. In response, researchers from LGC collaborated with manufacturers, proficiency-testing-scheme providers, and clinicians to develop the first commutable, matrix of clinical reference material for tacrolimus in whole blood. This reference material will improve confidence in measurement, irrespective of the analytical platform used and will enable clinicians to set and maintain optimal patient dosage to the benefit of their patients and the healthcare system. Experience gained here is now being translated into the development of a pure and matrix reference material for sirolimus, another immunosuppressant drug particularly used in liver transplants.
Diagnostic measurement challenges are rapidly progressing from small molecule analytes (e.g., cholesterol and glucose) toward larger, more analytically challenging and complex biomolecules (e.g., protein markers relevant for therapeutic intervention and some disease-state biomarkers). However, development of reference measurement procedures for these complex biomolecules is necessary to enable in vitro diagnostic and clinical measurement comparability. Due to their complexity and large size, achieving metrological traceability to the SI for protein biomarkers is not a trivial task.
In a major step forward, under the European Metrology Research Programme, several NMIs are collaborating to develop an international standardization framework for complex biomolecules. This research is focused on establishing reference measurement procedures capable of providing results that are traceable to the SI in order to generate a more thorough understanding of issues related to measurand description, reference material commutability, and the possible impact of establishing metrological traceability to the SI for protein biomarkers. Human growth hormone was used as a test case to investigate these new approaches.
Human growth hormone (hGH) is widely used in the diagnosis of disorders of children with short stature, management of disorders that lead to nutritional deficiency, and to monitor growth hormone replacement therapy. Therefore, there is a need for reliable and comparable measurements. In order to achieve this, routine measurement results need to be made traceable to a stable reference.
In order to begin to determine the concentration of a protein in serum, such as hGH, the protein was initially “broken down” into peptides. By choosing a number of peptide sequences unique to hGH, and by employing IDMS approaches, the concentration of the protein could then be accurately determined. In this case, the standards used were peptides and the concentration of the standard peptide solutions was determined by amino acid analysis (figure 2). This establishes a firm anchor to the amount of substance of the amino acids, while maintaining specificity to the hGH molecule, hence providing a unbroken link for the measurement results to the SI.
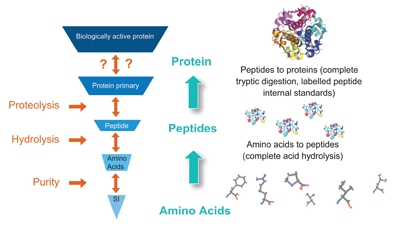 |
| Figure 2: Basic concept of the traceability for
protein IDMS methods. |
However, small differences in the primary structure of hGH may be expected due to differences in the genetic makeup of the host, such as the person in which the protein resides. These differences may give rise to a different chemical molecule due to the different sequence of amino acids. However, as many proteins are identified by function alone, the generic identity of the protein may remain unaltered. Consideration of this may be needed for the future production of commutable reference materials. In addition, transient changes to a protein’s secondary, tertiary, or quaternary structure may affect protein function and disease state. This means that it is not necessarily the total amount of the protein’s primary sequence that is important, but the amount of a protein in a particular structure or folding state. Many of the routine measurement methods used for protein biomarkers are immunoassay based where the specific binding of proteins can be structurally dependent. The key challenge for comparability of protein measurement results is not just the “amount of substance,” but the “function/activity” of the protein. There is therefore a need to develop an understanding of the effects that protein structure may have on the traceability of measurement results.
Advanced mass spectrometry-based techniques such as hydrogen deuterium exchange experiments and ion mobility coupled to mass spectrometry enable interrogation of protein structure under physiological conditions, thereby providing a measure of the different structural forms of the protein present. Monitoring the rate of exchange of hydrogen atoms present on a protein with deuterium provides information on the structural differences between different preparations of the same protein. Hydrogen atoms on the outside of the protein molecule will have a higher rate of exchange than hydrogen atoms in the protein folds, thereby providing a mechanism by which to follow structural change. This can be performed in the presence of specified ligands, enabling the determination of binding constants and the degree of activity. Ion mobility separates ions on the basis of collisional cross sections, which enable proteins of the same mass but different folding states to be separated. During the research, a number of different, standard protein preparations of recombinant hGH were investigated. Knowing the difference in protein structures, the relative amounts of these structures, and the interaction of these structural forms with detection antibodies will be an important factor in establishing the suitability of reference materials to standardize measurements.
The ultimate aim of standardization in clinical chemistry is to ensure comparability of routine measurements in order to achieve equivalence between results obtained by different methods. This research demonstrates the first steps towards developing a biometrology framework that aims to encompass both metrological rigor in developing SI traceability and the real concerns of the clinical community that measurements must be relevant and commutable, independent of the analytical platform used.
Dr. Gavin O’Connor <[email protected]> is principal scientist, Mass Spectrometry, LGC, Teddington, Middlesex, UK.
www.lgc.co.uk
References
- Note on terminology. The full term “metrological traceability” is used in international guides and standards to distinguish this concept from other forms of traceability such as traceability of samples, documents, or supply chains. In this article the term “traceability” should be understood as referring only to metrological traceability.
- ISO/IEC Guide 99-12:207, International Vocabulary of Metrology
|
LGC played its part in helping to celebrate the International Year of Chemistry 2011 by highlighting the achievements of chemistry and its contributions to the well-being of humankind.
 From hosting an open evening, during which interested members of the public were invited into the laboratories at its London based headquarters, to writing articles for the general scientific press and generating an online blog, LGC has spent 2011 spreading the word of chemistry to old and young, professional and non-professional alike. From hosting an open evening, during which interested members of the public were invited into the laboratories at its London based headquarters, to writing articles for the general scientific press and generating an online blog, LGC has spent 2011 spreading the word of chemistry to old and young, professional and non-professional alike.
LGC’s blog, LGC Science provides short, bite-sized articles highlighting how chemistry, in particular chemical measurement, impacts upon our everyday lives. It discusses some of LGC’s latest research and demonstrates how the company and its scientists apply their research to ensure “Science for a safer world.”
The LGC Science blog highlights some of the most exciting research projects, enlightens readers about what goes on behind the scenes in LGC’s forensics teams, discusses some of the cases solved through LGC’s unique Government Chemist function, and describes cutting edge research from other teams across LGC.
www.chemistry2011.org/participate/activities/show?id=1063
|
Page
last modified 3 January 2012.
Copyright © 2003-2012 International Union of Pure and Applied Chemistry.
Questions regarding the website, please contact [email protected] |